Top Ten Properties of Titanium
Low density, high specific strength
The density of metallic titanium is 4.51g/cm3, which is higher than aluminum and lower than steel, copper, and nickel, but the specific strength is the highest among metals.
Corrosion resistance
Titanium is a very active metal with a low equilibrium potential and a high tendency for thermodynamic corrosion in media. However, titanium is actually very stable in many media. For example, titanium is resistant to corrosion in media such as oxidative, neutral and weak reducibility. This is because titanium and oxygen have a great affinity. In air or oxygen-containing medium, a dense, strong and inert oxide film is formed on the surface of titanium to protect the titanium matrix from corrosion. Even if it is due to mechanical wear, it will quickly heal or regenerate. This shows that titanium is a metal with a strong tendency to passivation. The oxide film of titanium below the temperature of 315°C always maintains this characteristic.
In order to improve the corrosion resistance of titanium, surface treatment technologies such as oxidation, electroplating, plasma spraying, ion nitriding, ion implantation, and laser processing have been studied. Titanium oxide film played a role in enhancing protection and obtaining desired corrosion resistance. For the needs of metal materials in the production of sulfuric acid, hydrochloric acid, methylamine solution, high-temperature wet chlorine and high-temperature chloride, Titanium-molybdenum, titanium-palladium, titanium-molybdenum-nickel and other corrosion resistant titanium alloys have been developed. Titanium-32-molybdenum alloys are used in titanium castings. Titanium-0.3 molybdenum-0.8 nickel alloys are used in environments where crevice corrosion or pitting corrosion frequently occurs. Titanium-0.2-palladium alloys were used locally on titanium equipment, and good results were obtained.

Good heat resistance
The new titanium alloy can be used long-term at 600 °C or higher.
Low temperature resistance
The low-temperature titanium alloy represented by titanium alloys TA7 (Ti-5Al-2.5Sn), TC4 (Ti-6Al-4V) and Ti-2.5Zr-1.5Mo, etc., whose strength increases with decreasing temperature, However, plasticity is not great. It maintains good ductility and toughness at -196-253°C and avoids cold brittleness. It is an ideal material for cryogenic vessels and tanks.
Strong anti-damping performance
After metal titanium is subjected to mechanical vibration and electric vibration, its own vibration decay time is the longest compared with steel and copper metal. The use of this property of titanium can be used as a tuning fork, a medical ultrasonic vibrator vibration element, and an advanced sound speaker vibration film.
Non-magnetic, non-toxic
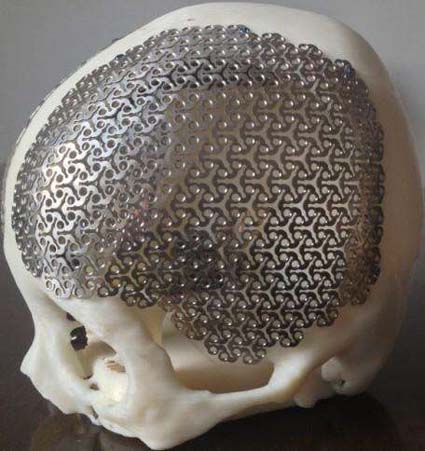
Titanium is a non-magnetic metal, it will not be magnetized in a large magnetic field, non-toxic and has good compatibility with human tissues and blood, so it is used by the medical community.
Tensile strength is close to its yield strength
This property of titanium shows that the yield strength ratio (tensile strength/yield strength) is high, indicating that the metal titanium material is poor in plastic deformation during forming. Since the ratio of the yield limit of titanium to the elastic modulus is large, the resilience of titanium when molded is large.
Good heat transfer performance
Although the thermal conductivity of metallic titanium is lower than that of carbon steel and copper, However, due to the excellent corrosion resistance of titanium, the wall thickness can be greatly reduced, and the heat exchange between the surface and the steam is drop-shaped condensation, reducing the thermal group. Titanium surface without scaling can also reduce the thermal resistance, so that the heat transfer performance of titanium is significantly improved.
Low modulus of elasticity
The elastic modulus of titanium is 106.4 GPa at room temperature, which is 57% of steel.
Suction performance
Titanium is a chemically very reactive metal that reacts with many elements and compounds at high temperatures. Titanium inhalation mainly refers to reaction with carbon, hydrogen, nitrogen, and oxygen at high temperatures.
The density of metallic titanium is 4.51g/cm3, which is higher than aluminum and lower than steel, copper, and nickel, but the specific strength is the highest among metals.
Corrosion resistance
Titanium is a very active metal with a low equilibrium potential and a high tendency for thermodynamic corrosion in media. However, titanium is actually very stable in many media. For example, titanium is resistant to corrosion in media such as oxidative, neutral and weak reducibility. This is because titanium and oxygen have a great affinity. In air or oxygen-containing medium, a dense, strong and inert oxide film is formed on the surface of titanium to protect the titanium matrix from corrosion. Even if it is due to mechanical wear, it will quickly heal or regenerate. This shows that titanium is a metal with a strong tendency to passivation. The oxide film of titanium below the temperature of 315°C always maintains this characteristic.
In order to improve the corrosion resistance of titanium, surface treatment technologies such as oxidation, electroplating, plasma spraying, ion nitriding, ion implantation, and laser processing have been studied. Titanium oxide film played a role in enhancing protection and obtaining desired corrosion resistance. For the needs of metal materials in the production of sulfuric acid, hydrochloric acid, methylamine solution, high-temperature wet chlorine and high-temperature chloride, Titanium-molybdenum, titanium-palladium, titanium-molybdenum-nickel and other corrosion resistant titanium alloys have been developed. Titanium-32-molybdenum alloys are used in titanium castings. Titanium-0.3 molybdenum-0.8 nickel alloys are used in environments where crevice corrosion or pitting corrosion frequently occurs. Titanium-0.2-palladium alloys were used locally on titanium equipment, and good results were obtained.

Good heat resistance
The new titanium alloy can be used long-term at 600 °C or higher.
Low temperature resistance
The low-temperature titanium alloy represented by titanium alloys TA7 (Ti-5Al-2.5Sn), TC4 (Ti-6Al-4V) and Ti-2.5Zr-1.5Mo, etc., whose strength increases with decreasing temperature, However, plasticity is not great. It maintains good ductility and toughness at -196-253°C and avoids cold brittleness. It is an ideal material for cryogenic vessels and tanks.
Strong anti-damping performance
After metal titanium is subjected to mechanical vibration and electric vibration, its own vibration decay time is the longest compared with steel and copper metal. The use of this property of titanium can be used as a tuning fork, a medical ultrasonic vibrator vibration element, and an advanced sound speaker vibration film.
Non-magnetic, non-toxic
Titanium is a non-magnetic metal, it will not be magnetized in a large magnetic field, non-toxic and has good compatibility with human tissues and blood, so it is used by the medical community.
Tensile strength is close to its yield strength
This property of titanium shows that the yield strength ratio (tensile strength/yield strength) is high, indicating that the metal titanium material is poor in plastic deformation during forming. Since the ratio of the yield limit of titanium to the elastic modulus is large, the resilience of titanium when molded is large.
Good heat transfer performance
Although the thermal conductivity of metallic titanium is lower than that of carbon steel and copper, However, due to the excellent corrosion resistance of titanium, the wall thickness can be greatly reduced, and the heat exchange between the surface and the steam is drop-shaped condensation, reducing the thermal group. Titanium surface without scaling can also reduce the thermal resistance, so that the heat transfer performance of titanium is significantly improved.
Low modulus of elasticity
The elastic modulus of titanium is 106.4 GPa at room temperature, which is 57% of steel.
Suction performance
Titanium is a chemically very reactive metal that reacts with many elements and compounds at high temperatures. Titanium inhalation mainly refers to reaction with carbon, hydrogen, nitrogen, and oxygen at high temperatures.






